The theory of particle filtration is well documented in literature and has been verified in practice down to lowest detectable particle sizes of available measuring equipment (Lee and Liu, 1980).
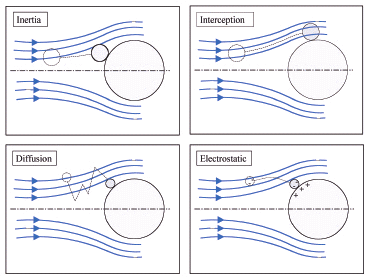
A filter's ability to capture particles depends on different physical and mechanical phenomena including diffusion, interception as well as inertial and sieving effects. Electrostatic interactions between fibres and the collected particles can also be of decisive importance, particularly in the case of synthetic polymer fibres which can carry high levels of electrostatic charge. In an ideal filtration process, each particle entering the filter is permanently arrested on initial contact with a fibre or a particle already captured. When small particles are filtered at low air velocities, the adhesive forces (van der Waal forces) greatly exceed the kinetic energy of the airborne particles and such particles, once captured, are unlikely to be dislodged from the filter. As particle size and air velocity increase, “bouncing off" of larger particles from fibres becomes a more likely occurrence.
Particles which are larger in diameter than the clearance between two fibres cannot pass. But the mainly existing assumption, that air filters act like a sieve is fundamentally wrong, as stated by the following filtration effects.
Large particles have high momentum which makes it more difficult for them to follow the air stream around a fibre. The particles continue on their original path, contact the fiber and remain there. These inertial effects increase with increased air velocity as well as increased size or weight of the particle. Accordingly the inertial effect is the predominant separation mechanism for particles > 1µm.
Small particles follow the air flow around the fibre and are captured if they come close enough to the fiber. The effect increases with increased particle size, smaller fibre diameters and/or smaller clearance between (i.e. closer spacing of) the fibres.
Diffusion in the lengthwise direction of media fibres can be found only in the case of very small particles such as viruses. The particles move randomly about within the air stream due to Brownian molecular motion. This mechanism is only important for particles with diameters < 1µm. The lower the air flow rate during operation of an air filter, the higher the efficiency levels against tiny submicron particles, as they have more time for a diffusive separation. Once a submicron particle has been captured on a fibre, it is hardly possible for the particle to seperate again, as immense, unrealistically forces would be required during a regular operation.
With what filtration effect do heavy and large particles make their own way to contact with the fibers of the filter medium?